Quantitative Carrier Test Evaluating Virucidal Activity of Chemical Disinfectants on Non-porous Surfaces
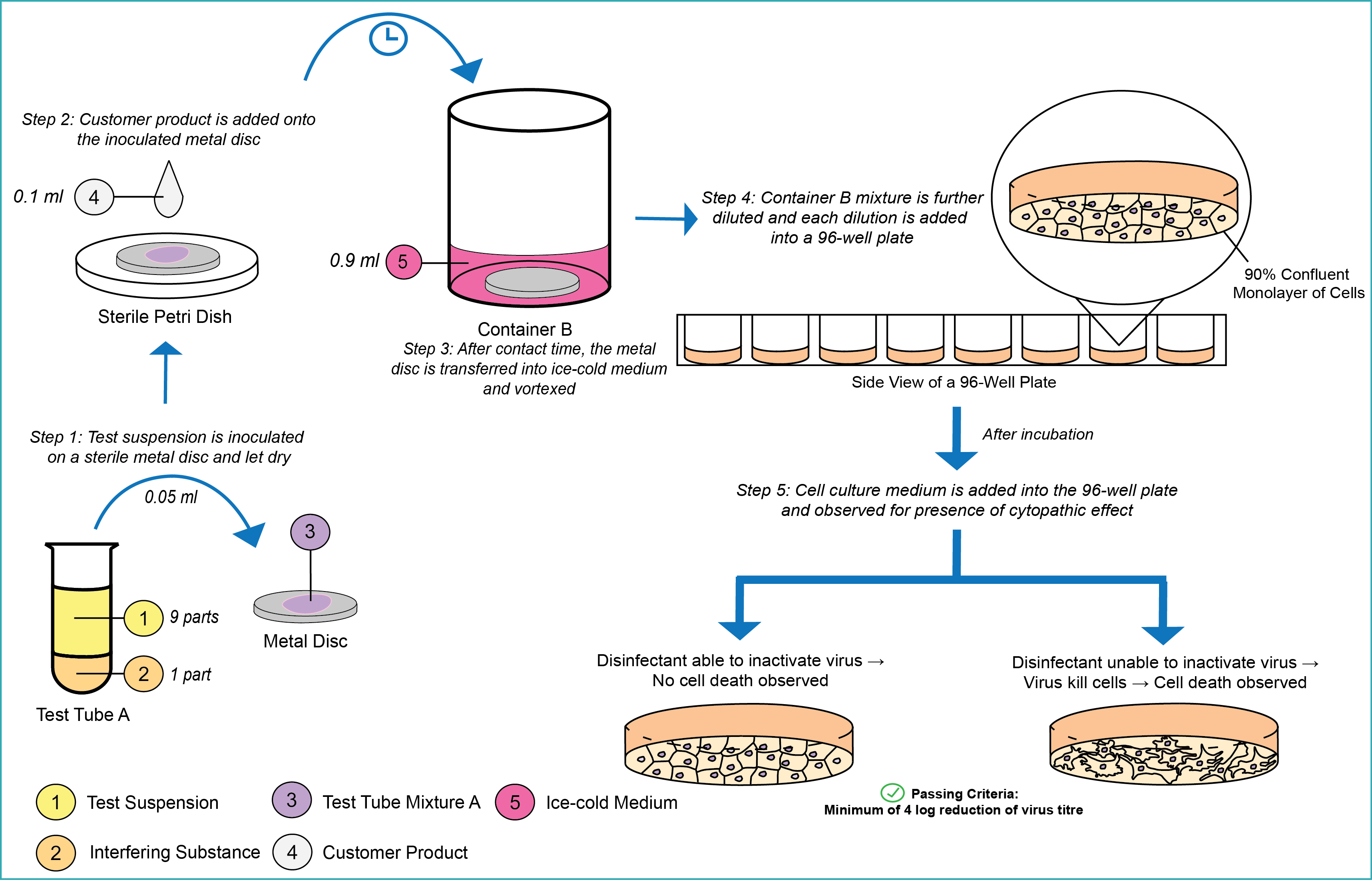
EN 16777 is an essential European Standard used to evaluate the minimum virucidal activity requirements of surface disinfectants used on non-porous surfaces in the medical area. The infographic below shows a brief insight of EN 16777.
CUSTOMER PROCESS
Step 1: Is your product a surface disinfectant?
This test is suitable to evaluate surface disinfectants that require no wiping action used in the medical area.
Step 2: Select your test microorganisms
These are the mandatory virus strains for testing according to virucidal activity claims:
Virucidal Activity:
- Adenovirus
- Murine norovirus
Δ The product shall pass against Poliovirus, Adenovirus and Murine Norovirus in EN 14476 prior to EN 16777
Limited Spectrum Virucidal Activity:
- Adenovirus
- Murine norovirus
Δ The product shall pass against Adenovirus and Murine Norovirus in EN 14476 prior to EN 16777
Virucidal Activity Against Enveloped Virus:
- Vaccinia virus
Δ The product shall pass against Vaccinia virus in EN 14476 prior to EN 16777
Wish to add more microorganisms? Click here for our full list of microorganisms.
Step 3: Select an appropriate contact time and concentration for your test product
The test concentration is chosen according to the manufacturer’s recommendation.
The contact time recommended by the test method is no longer than 5 minutes for surfaces frequently touched by different people, and no longer than 60 minutes for other surfaces.
Step 4: What are the soiling conditions associated with your test product?
Interfering substances, or commonly referred to as soiling, are used to mimic organic substances found on human skin, surfaces, and instruments. These are the available soiling conditions according to the test method:
- Clean condition: 0.3 g/L bovine serum albumin
- Dirty condition: 3.0 g/L bovine serum albumin + 3.0 mL/L sheep erythrocytes
Step 5: Place an order and send your sample to us
Fill in the test request form and place an order via email at info@tecolab-global.com. We will advise on the quantity of sample required for testing.
You can deliver the sample to us via courier and provide us the tracking code. We will monitor the delivery status and inform you when we receive the sample.
Step 6: Track the status of your request
Step 7: Download your password-protected test report
Download your readily available test report online with just a single click using the link and password provided via email notification. We will receive a notification once you have downloaded the test report.
For safety purposes, the uploaded test report will self-destruct after 7 days.
Step 8: Analyze your passing criteria in the test report with our laboratory experts
In order to pass the test, the product shall demonstrate at least a 4 decimal log (lg) reduction.
Need assistance with your test report? Discuss your results with our microbiologists for more information.


 Deutsch
Deutsch
 中文
中文
 日本語
日本語
 Português
Português
 Français
Français
 Español
Español
 العربية
العربية